04.05.2020 by Claire Strasser
Thermal Stability of Drugs
The Thermal StabilityA material is thermally stable if it does not decompose under the influence of temperature. One way to determine the thermal stability of a substance is to use a TGA (thermogravimetric analyzer). thermal stability of a substance is closely related to the shelf-life of pharmaceutical products. Here, the Thermal StabilityA material is thermally stable if it does not decompose under the influence of temperature. One way to determine the thermal stability of a substance is to use a TGA (thermogravimetric analyzer). thermal stability of diclofenac sodium (anti-inflammatory) is determined by means of thermogravimetry.
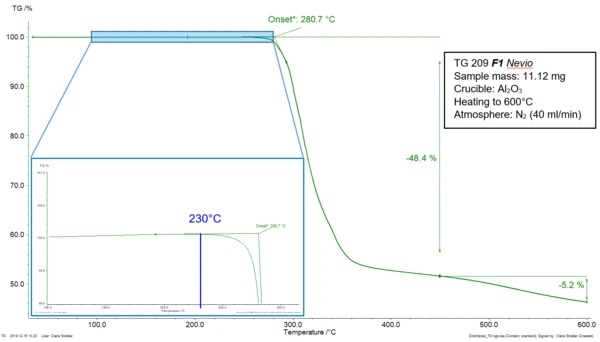
A material is thermally stable as long as no change occurs in its structure and properties when submitted to higher temperatures. Standard ASTM E2550-17 describes the thermal stability of a substance as “the temperature at which [it starts] to decompose or react” [1] during a thermogravimetric measurement. In this technique, the influence of the temperature on the behavior of a sample is determined. For that, “the mass of a substance is measured as a function of temperature or time while the substance is subjected to a controlled-temperature program in a specified atmosphere.” [2] Figure 1 depicts the TGA measurement of diclofenac sodium during heating at 10 K/min in a nitrogen atmosphere. The mass-loss step of the curve can be characterized by the extrapolated onset temperature at 281°C as described in ISO 11358-1 [3]. In ASTM E2550-17, the beginning of the step is determined by “the point on the TG curve where a deflection is first observed from the established baseline prior to the thermal event.” [1] In the given example, it is located at 230°C (blue indication in the zoomed area). The mass loss occurring at 281°C (extrapolated onset temperature, ISO 11358-1) and 230°C (onset temperature, ASTM E2550-17), is related to the thermal Decomposition reactionA decomposition reaction is a thermally induced reaction of a chemical compound forming solid and/or gaseous products. decomposition of the sample.
You will find more information about Thermal StabilityA material is thermally stable if it does not decompose under the influence of temperature. One way to determine the thermal stability of a substance is to use a TGA (thermogravimetric analyzer). thermal stability measurements here.